AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Water in oil emulsion volum fraction11/28/2023  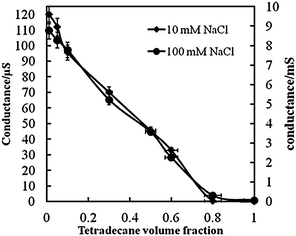
Some types of nanoemulsions have been shown to effectively destroy HIV-1 and various tuberculosis pathogens, for example, on non-porous surfaces.Īn emulsifier (also known as an emulgent) is a substance which stabilizes an emulsion, frequently a surfactant. The most effective application of this type of nanoemulsion is for the disinfection of surfaces. For this reason, nanoemulsions of this type are not yet ready to be used intravenously. The exceptions are sperm cells and blood cells, which are vulnerable to nanoemulsions due to their membrane structures. Remarkably, the soybean oil emulsion does not harm normal human cells nor the cells of most other higher organisms. On a mass scale, this effectively disintegrates the membrane and kills the pathogen. The oil is emulsified with detergents to stabilize the emulsion (the droplets won't merge with one another), so when they encounter lipids on a bacterial membrane or a virus envelope, they force the lipids to merge with themselves. The smaller the droplet, the greater the surface tension and thus the greater the force to merge with other lipids. The process is not chemical, as with other types of anti-pathogenic treatments, but physical. In medicine, a nanoemulsion of soybean oil to create drops of 400-600 nanometers in diameter will kill many pathogens such as bacteria and viruses. Nanoemulsion is a type of emulsion in which the sizes of the particles in the dispersed phase are defined as less than 1000 nanometers. There are three types of emulsion instability: flocculation, where the particles form clumps creaming, where the particles concentrate towards the surface (or bottom, depending on the relative density of the two phases) of the mixture while staying separated and breaking and coalescence where the particles coalesce and form a layer of liquid.Įmulsion is also a term used in the oil field as untreated well production that consists primarily of crude oil and water. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion tends to imply that both the dispersed and the continuous phase are liquid. Fluid emulsions can also suffer from creaming, the migration of one of the substances to the top of the emulsion under the influence of buoyancy or centripetal force when a centrifuge is used.Įmulsions are part of a more general class of two-phase systems of matter called colloids. This phenomenon is called coalescence, and happens when small droplets recombine to form bigger ones. Homemade oil and vinegar salad dressing is an example of an unstable emulsion that will quickly separate unless shaken continuously. Surface active substances ( surfactants) can increase the kinetic stability of emulsions greatly so that, once formed, the emulsion does not change significantly over years of storage. Over time, emulsions tend to revert to the stable state of oil separated from water. Energy input through shaking, stirring, homogenizers, or spray processes are needed to form an emulsion. Emulsions are unstable and thus do not form spontaneously. Emulsification is the process by which emulsions are prepared.Įmulsions tend to have a cloudy appearance, because the many phase interfaces (the boundary between the phases is called the interface) scatter light that passes through the emulsion. In milk and cream, oil is dispersed within a continuous water phase (an oil-in-water emulsion). In butter and margarine, a continuous liquid phase surrounds droplets of water (a water-in-oil emulsion). Examples of emulsions include butter and margarine, milk and cream, espresso, mayonnaise, the photo-sensitive side of photographic film, and cutting fluid for metal working. One substance (the dispersed phase) is dispersed in the other (the continuous phase). An emulsion is a mixture of two immiscible (unblendable) substances. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
